Phosphorylated phosphoinositides (PIPs) are phospholipids located at specific cellular membranes. Different phosphorylated PIs can be specifically recognized by their effectors, which will then be recruited to PI-enriched membranes. Phosphatidylinositol 3,5-bisphosphate (PI(3,5)P2) is an essential phosphoinositide required for endosome homeostasis and sorting for lysosomal degradation. However, the underlying mechanisms, especially in mammals, remain elusive or unexplored.
A recent study carried out by scientists from Drs. LIU Jinsong and SHU Xiaodong’s groups of Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences (GIBH.CAS) determined a structure of PI(3,5)P2 bound to Sorting Nexin 11 (SNX11), a protein contains a PX domain, with an opened PPII-C loop and obtained a SNX11 structure with its PPII-C in “closed” form that serves as a potential PI3P-binding model. This study, for the first time, provides details of how PX domain-containing proteins recognize the di-phosphorylated PI in the PI-associated sorting processes.
In addition, the study reveals that vacuolar H+-ATPase (V-ATPase) can interact with SNX11, which provides a link between PI(3,5)P2 and human V-ATPase and further evidence for their roles in the endosome homeostasis regulation. Lastly, combined with MD studies, the study shows that the α5 helix can unfold from the PX domain of SNX11 when targeting the membrane or interacting with its partner. These findings identify a novel PI(3,5)P2 effector, which will shed light on the PIs recognizing mechanism and the understanding of the downstream sorting events triggered by different PI binding.
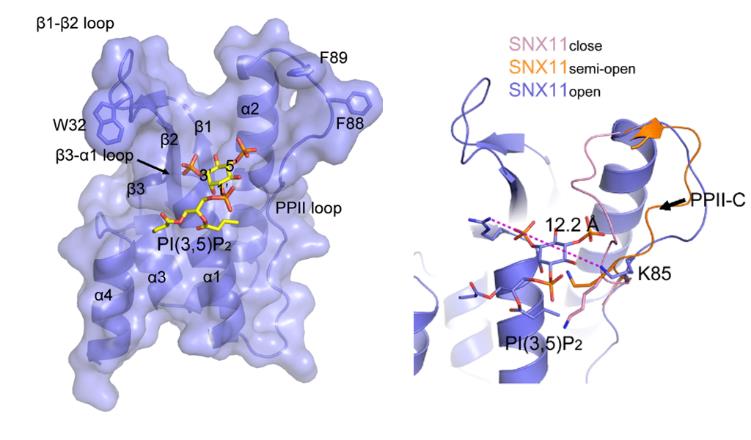
Figure. Surface representation of the complex structure of SNX11 with PI(3,5)P2 (left) and structural comparison among different states of the PPII-C loop of SNX11. (Image by XU Tingting)
Contact:
Dr. LIU Jinsong
Principal Investigator
Guangzhou Institute of Biomedicine and Health (GIBH), Chinese Academy of Sciences
E-mail: liu_jinsong@gibh.ac.cn
Keyword:SNX11, PI(3,5)P2, PI3P, lysosomal degradation, endosome homeostasis
 download:
download: