 Qiang Zhu, Ph.D. Qiang Zhu, Ph.D.
Principal Investigator
Dr. Qiang Zhu received his B.S. from the department of chemistry and chemical engineering, Nanjing University, in 1995 and Ph.D. from Shanghai Institute of Organic Chemistry (SIOC), Chinese Academy of Sciences (CAS) in 2000. He did his post-doctoral research at Colorado State University from 2000 to 2001. Then, he worked as a staff scientist for anti-HCV drug discovery at Vivoquest Inc. from 2011 to 2005 and at XTL Biopharmaceuticals Inc from 2005 to 2008, respectively. Dr. Zhu has been a principal investigator (PI) at Guangzhou Institutes of Biomedicine and Health (GIBH), CAS, since May 2008. He leads a group focusing on methodology development on the synthesis of heterocycles.
Personnel:

Research:
At GIBH, Dr. Zhu’s research interest focuses on the following areas: 1) Transition-metal-catalyzed C?H functionalization in the synthesis of heterocyclic compounds; 2) Copper-catalyzed dioxygen activation in the selective oxidation of small molecules; and 3) “Leads” identification and their structural modification for the purpose of anti-TB and anti-lung cancer drug discovery.
For example, the group developed a novel and efficient synthesis of pyrido[1,2-a]benzimidazoles through direct intramolecular aromatic C-H amination of N-aryl-2-aminopyridines. The reaction, co-catalyzed by Cu(OAc)2 and Fe(NO3)3?9H2O, is carried out in DMF under a dioxygen atmosphere. Diversified pyrido[1,2-a]benzimidazoles containing various substitution patterns are obtained in moderate to excellent yields by using this procedure. The results of mechanistic studies suggest that a Cu(III)-catalyzed electrophilic aromatic substitution (SEAr) pathway is operating in this process. The unique role of iron(III) is believed to lie in its ability to facilitate formation of the more electrophilic Cu(III) species. In the absence of iron(III), a much less efficient and reversible Cu(II)-mediated SEAr process takes place. This result was published in J. Am. Chem. Soc., a top international chemistry journal. 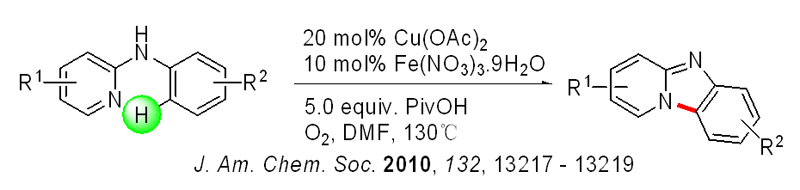
Another study has led to the development of an unprecedented intramolecular dehydrogenative aminooxygenation (IDA) process that produces imidazo[1,2-a] pyridine-3-carbaldehydes and 1,2-disubstituted imidazole-4-carbaldehydes from readily available N-allyl-2-aminopyridines and substituted N-allylamidines, respectively. The reaction, carried out by using 20 mol % CuII catalyst in DMF or DMA under a dioxygen atmosphere, is efficient and environmentally benign since it does not require additional organic or inorganic oxidants. Substituted imidazo[1,2-a]pyridine-3-carbaldehydes and imidazole-4-carbaldehydes, which can serve as versatile synthetic intermediates, are obtained in moderate to good yields by a reaction that possesses a broad substrate scope and good functionality tolerance. The process opens a new way towards direct formation of formyl-group substituted aromatic N-heterocycles from acyclic substrates. Mechanistic studies directly show that the carbonyl oxygen in the aldehyde products is derived from dioxygen by a pathway that takes place via a peroxycopper(III) intermediate. 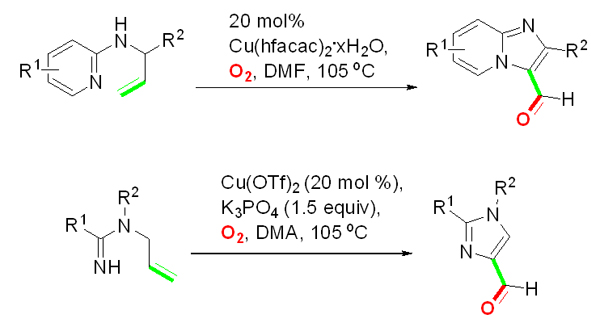
Publications: | 1. | Yong Wang, Honggen Wang, Jiangling Peng, Qiang Zhu*
Palladium-Catalyzed Intramolecular C(sp2)–H Amidination by Isonitrile Insertion Provides Direct Access to 4-Aminoquinazolines from N-Arylamidines
Org. Lett. 2011, ASAP. | | 2. | Honggen Wang, Yong Wang, Dongdong Liang, Lanying Liu, Jiancun Zhang,* Qiang Zhu*
Copper-Catalyzed Intramolecular Dehydrogenative Aminooxygenation: Direct Access to Formyl-Substituted Aromatic N-Heterocycles
Angew. Chem. Int. Ed. 2011, 50, 5678-5681. | | 3. | LijiecPeng, Honggen Wang, Changlan Peng, Ke Ding,* Qiang Zhu*
Sequential Hydration–Condensation–Double Cyclization of Pyridine-Substituted 2-Alkynylanilines: An Efficient Approach to Quinoline-Based Heterocycles
Synthesis 2011, 1723-1732. | | 4. | Bin Ma, Yong Wang, Jiangling Peng, Qiang Zhu*
Synthesis of Quinazolin-4(3H)-ones via Pd(II)-Catalyzed Intramolecular C(sp2)-H Carboxamidation of N-arylamidines
J. Org. Chem. 2011, 76, 6362-6366. | | 5. | Changlan Peng, Yong Wang, Lanying Liu, Honggen Wang, Jiaji Zhao, Qiang Zhu*
p-Toluenesulfonic Acid Promoted Annulation of 2-Alkynylanilines with Activated Ketones: Efficient Synthesis of 4-Alkyl-2,3-Disubstituted Quinolines
Eur. J. Org. Chem. 2010, 818-822. | | 6. | Honggen Wang, Yong Wang, Changlan Peng, Jiancun Zhang,* and Qiang Zhu*.
A Direct Intramolecular C-H Amination Reaction Cocatalyzed by Copper(II) and Iron(III) as Part of an Efficient Route for the Synthesis of Pyrido[1,2-a]benzimidazoles from N-Aryl-2-aminopyridines
J. Am. Chem. Soc. 2010, 132, 13217-13219. | | 7. | Yong Wang, Hong Lu, Qiang Zhu,* Shibo Jiang,* Yun Liao*
Structure-Based Design, Synthesis and Biological Evaluation of New N-Carboxyphenylpyrrole Derivatives as HIV Fusion Inhibitors Targeting gp41
Bioorg. Med. Chem. Lett. 2010, 20, 189-192. | | 8. | Jiaji Zhao, Changlan Peng, Lanying Liu, Yong Wang, and Qiang Zhu*.
Synthesis of 2-Alkoxy(aroxy)-3-substituted Quinolines by DABCO-Promoted Cyclization of o-Alkynylaryl Isocyanides
J. Org. Chem. 2010, 75, 7502-7504. | | 9. | Lanying Liu, Yong Wang, Honggen Wang, Changlan Peng, Jiaji Zhao, Qiang Zhu*
Tetrabutylammonium Chloride-Triggered 6-endo Cyclization of o-Alkynylisocyanobenzenes: an Efficient Synthesis of 2-Chloro-3-Substituted Quinolines
Tetrahedron Letters 2009, 50, 6715-6719. | | 10. | Honggen Wang, Lanying Liu, Yong Wang, Changlan Peng, Jiancun Zhang,* Qiang Zhu*
An efficient Synthesis of 4-Alkyl-2(1H)-Quinazolinones and 4-Alkyl-2-Chloroquinazolines from 1-(2-Alkynylphenyl)ureas
Tetrahedron Letters 2009, 50, 6841-6843. | | 11. | Yong Wang, Changlan Peng, Lanying Liu, Jiaji Zhao, Li Su, Qiang Zhu *.
Sulfuric Acid Promoted Condensation Cyclization of 2-(2-(Trimethylsilyl) ethynyl)anilines with Arylaldehydes in Alcoholic Solvents: an Efficient One-pot Synthesis of 4-Alkoxy-2-Arylquinolines
Tetrahedron Letters 2009, 50, 2261-2265. |
|