Researchers reveal a cell-fate "rheostat" governing somatic reprogramming
A recent study from Professor LIU Jing’s team at the Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences (GIBH, CAS), was published online in Stem Cell Reports as “Dose-Resolved Control of Somatic Reprogramming by Rora”and was accompanied by an invited Preview by Professor Constantinos Chronis in the same issue. The study reports that the nuclear receptor RORA functions as a dose-tunable “rheostat” in OKS-mediated somatic reprogramming: moderate Rora expression enhances iPSC colony formation, whereas high Rora expression suppresses stable colony formation, revealing a biphasic dose response.
To systematically assess nuclear receptor functions in reprogramming, the researchers screened 49 murine nuclear receptors and identified the ROR subfamily as consistent enhancers, with Rora showing the strongest effect. Domain analyses further indicated that the DNA-binding and ligand-binding domains are required for the pro-reprogramming activity, whereas the N-terminal domain contributes to inhibition at high expression.
Mechanistically, integrated multi-omics analyses linked low-dose Rora to attenuation of IFN-γ signaling (and IFN-γ add-back dampened the enhancement), while high-dose Rora was associated with reduced WNT pathway output; activating WNT signaling with CHIR99021 partially rescued the high-dose defect..
Overall, this study supports the concept that transcription factors can function as “rheostats” rather than simple switches, and provides a dosage-based framework for improving iPSC reprogramming and cell-fate control.
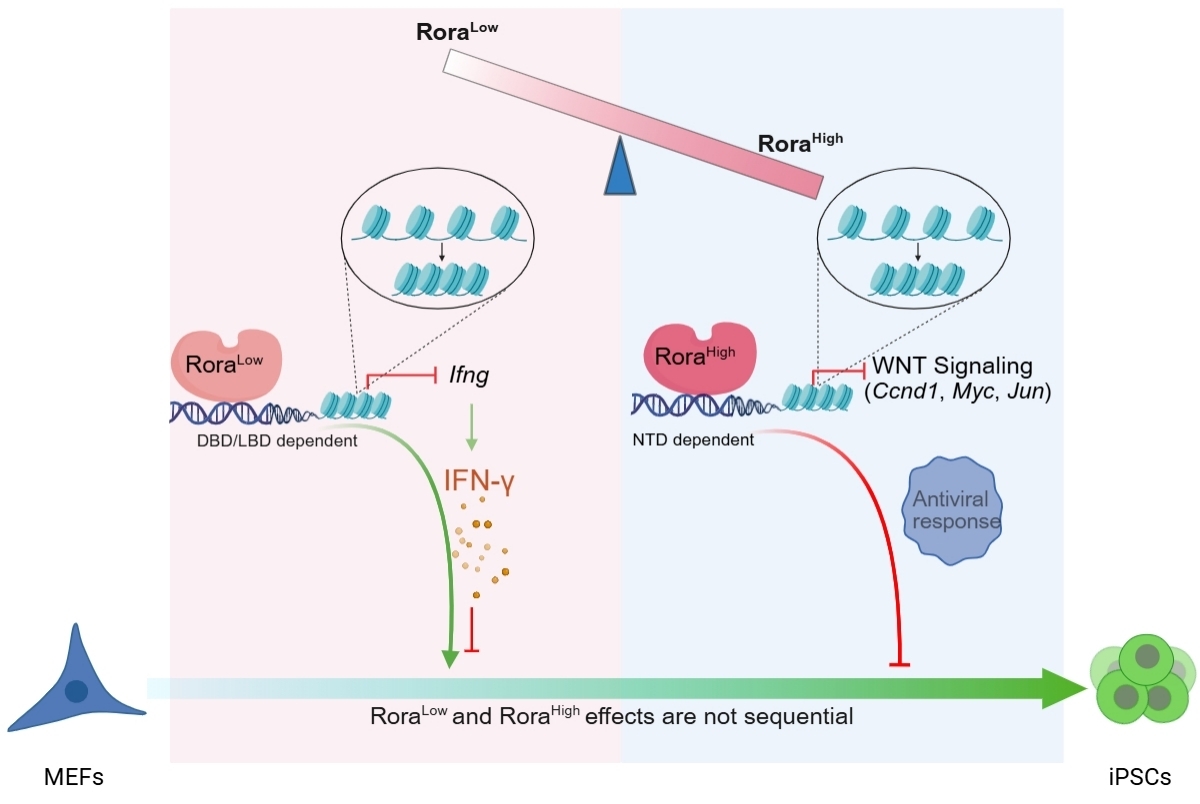 A proposed model of RORA's biphasic dose effect in somatic reprogramming
A proposed model of RORA's biphasic dose effect in somatic reprogramming
Images provided by Professor LIU’s team
This work was completed in collaboration with the Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences, and the Centre for Regenerative Medicine and Health, Hong Kong Institute of Science & Innovation, Chinese Academy of Sciences. Professor LIU Jing and Dr. Manish Kumar are the co-corresponding authors; WANG Haiyun, LI Yusha, and YIN Chunkou are co-first authors.
Contacts:
LIU Jing, Ph.D., Principal Investigator
Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences, Guangzhou, China, 510530.
Email: liu_jing@gibh.ac.cn
Attachment Download:
-
ContactLIU Jing, Ph.D.liu_jing@gibh.ac.cn
-
Reference
-
Related Article