GIBH Collaborates to Develop Selective DNMT1 Inhibitors for Hypomethylating Agent-Resistant Acute Myeloid Leukemia
A collaborative team from the Guangzhou Institutes of Biomedicine and Health (GIBH), Chinese Academy of Sciences, Johns Hopkins University, and the Hangzhou Institute for Advanced Study recently published a study in PNAS entitled Structure-guided design of 7-azaindole DNMT1 inhibitors active against hypomethylating agent–resistant acute myeloid leukemia. The study describes a class of DNA methyltransferase 1 (DNMT1) inhibitors with a 7-azaindole core scaffold. A representative compound, DMI46, exhibited potent therapeutic activity in models of hypomethylating agent (HMA)-resistant acute myeloid leukemia (AML), supporting its potential as a candidate therapy for refractory AML.
Promoter DNA hypermethylation can silence key tumor suppressor genes (TSGs) and is a hallmark of human cancer, serving as a major epigenetic driver of tumor initiation and progression. Reversing aberrant DNA methylation to restore TSG expression via DNMT1 inhibition has emerged as an important therapeutic paradigm. Currently, the nucleoside HMAs decitabine and azacitidine are approved frontline therapies for hematologic malignancies such as AML. However, these agents must be incorporated into DNA to form DNMT1-DNA adducts, highly cytotoxic DNA lesions that cause dose-limiting toxicities and constrain the clinical benefit of first-generation HMAs. Overall response rates remain ~40%, and most initial responders acquire drug resistance and relapse within two years. There is therefore a pressing need for DNMT1 inhibitors (DNMT1i) with improved safety profiles and the ability to overcome resistance to nucleoside HMAs.
The team previously reported that conformational transitions of the DNMT1 catalytic domain between open and closed states are required for methyltransferase activity (ACS Chem Biol, 2018), suggesting the feasibility of developing non-nucleoside DNMT1i that block these transitions. In the present study, the researchers combined structure-guided scaffold hopping with chemical optimization to target this key conformational regulatory region. Using their sensitive cell-based screening platform for HMAs (Adv Sci, 2026), they identified a series of DNMT1i featuring a bicyclic 7-azaindole scaffold. Among these, DMI46 effectively reversed cancer-specific DNA hypermethylation, reactivated numerous epigenetically silenced TSGs, and exhibited strong anti-AML activities. Unlike decitabine, DMI46 induces minimal DNA damage and displays a markedly improved safety profile with a wider therapeutic window.
Cryo-EM analyses further revealed that the 7-azaindole scaffold not only forms key contacts with DNMT1 but also intercalates into DNA through enhanced π-π stacking, thereby anchoring the compound within the DNMT1-DNA complex. This configuration facilitates extension of the phenyl substituents toward the catalytic loop and DNA-recognition helix, locking DNMT1 in an open, inactive conformation and preventing its conformational transitions. These structural features underpin sustained on-target DNMT1 inhibition and robust antiproliferative activity of DMI46 in AML models resistant to either non-nucleoside or nucleoside HMAs.
Beyond acting on tumor cells, pharmacologic modulation of DNA methylation can also influence immune and hematopoietic cell fates, suggesting broader translational opportunities for 7-azaindole-based DNMT1i. These include chemical reprogramming approaches for adoptive immune cell therapies and combination strategies with immune checkpoint blockade to enhance antitumor efficacy in solid tumors. Moreover, in collaboration with Dr. Lan’s group at Fudan University, the team found that DMI46 selectively induces DNA demethylation and fetal hemoglobin activation, promotes erythroid maturation and alleviates β-thalassemia progression (Adv Sci, 2025), supporting potential utility in β-thalassemia, a highly prevalent genetic disorder in southern China.
Dr. TANG Shibing, ZONG Liangyi and SHANG Yini(GIBH), MA Shuyuan and WEI Jiale (Hangzhou Institute for Advanced Study) are co-first authors. The project was jointly supervised by Dr. KONG Xiangqian, Dr. Stephen Baylin, Dr. LI Zhihai and Dr. TANG Shibing, with contributions from Dr. LUO Cheng (Shanghai Institute of Materia Medica), Dr. CHEN Qi, Dr. PAN Guangjin and Dr. HUANG Jing (GIBH), Dr. YE Fei (Zhejiang Sci-Tech University), and Dr. WENG Hengyou (Guangzhou National Laboratory). The authors acknowledge financial support from the National Key R&D Program of China, the Chinese Academy of Sciences Strategic Priority Research Program, the GIBH Major Research Project, and the Major Research Program of Guangzhou National Laboratory.
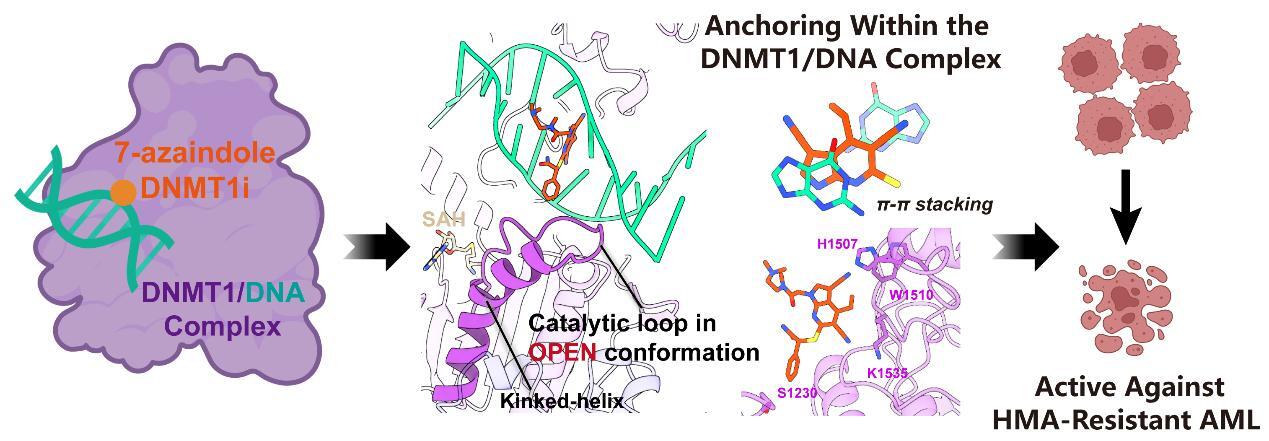
Figure. DMI46 traps DNMT1 in an open, inactive conformation and confers robust therapeutic activity in AML models resistant to current HMAs.
Contacts:
KONG Xiangqian, Ph.D., Principal Investigator
Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences
Guangzhou,China, 510530
Email: kong_xiangqian@gibh.ac.cn
Attachment Download:
-
ContactKONG Xiangqian, Ph.D.kong_xiangqian@gibh.ac.cn
-
Reference
-
Related Article