Researchers reveal organ-specific vascular development in embryonic mouse
A joint team from the Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences (GIBH, CAS), South China University of Technology, Guangzhou National Laboratory, and Westlake University published a study in Cell entitled A single-cell time-series atlas of endothelial cell embryonic development.
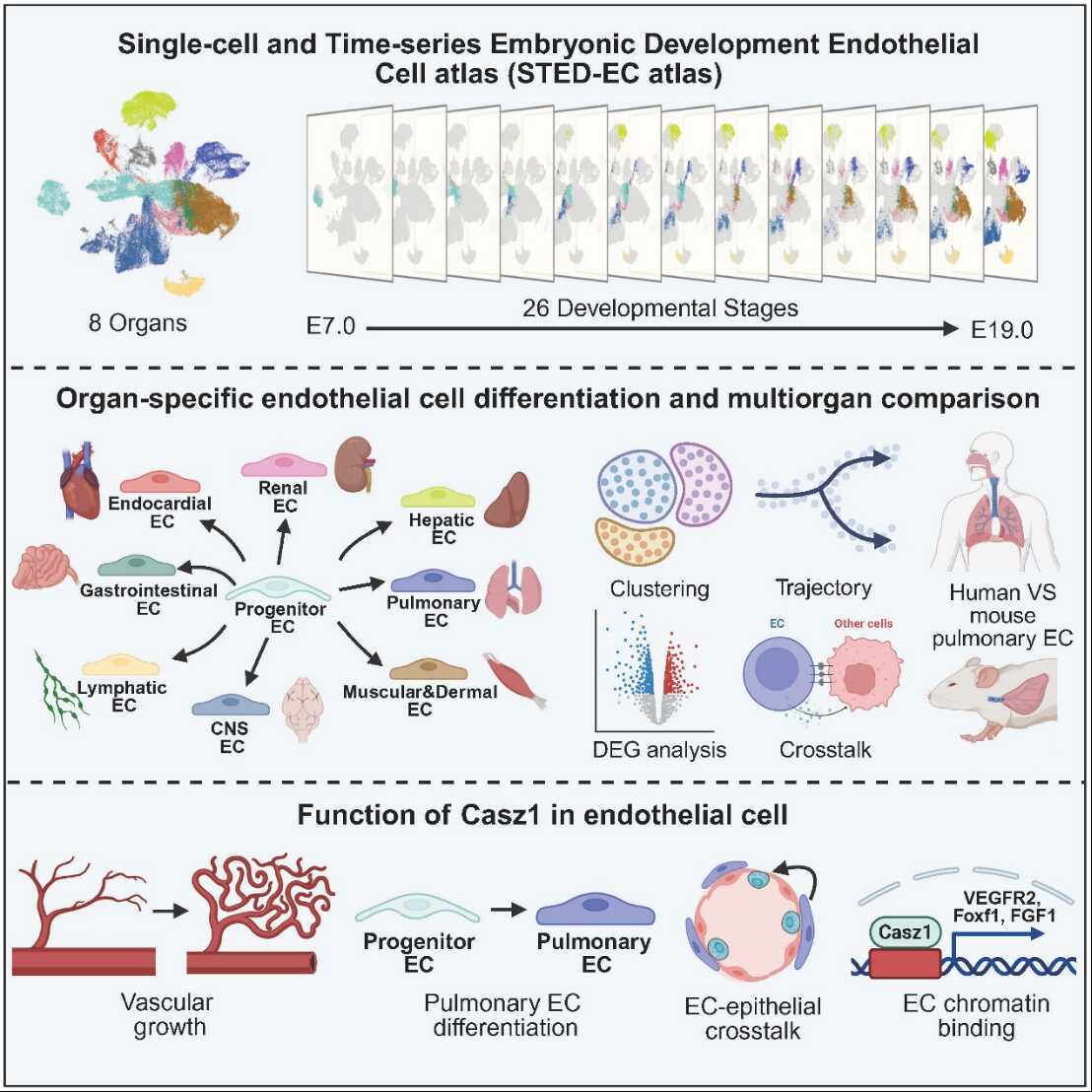
Through establishing a mouse endothelial development atlas covering entire embryonic stages (http://sted-ec.ccla.ac.cn/), this study shows that endothelial cells start to undergo organ-specific differentiation from mid-gestation. It also identifies Casz1 as a lung-enriched endothelial gene that supports lung vessel growth, organ-specific endothelial maturation, and communication between endothelial and epithelial cells in the lung.
Blood vessels extend throughout the vertebrate body. Beyond carrying blood, they help coordinate tissue repair and regeneration. Endothelial cells line the vessel interior and form the core structural layer of blood vessels. Although the arterial and venous endothelial differentiation has been studied in detail, how endothelial cells acquire organ-specific identities—and why this matters physiologically—remains unclear.
To address this question, the researchers established an endothelial developmental atlas covering the entire mouse embryonic period, with data including 8 organs and 26 time points. Endothelial cells could be identified by embryonic day 7 (E7). The organ-specific differentiation of endothelial cells initiated as early as E8 with cardiac endothelial cells (endocardium), and expanded during mid-gestation (E9.0–E13.5) across multiple organs, including the central nervous system, liver, lung, and digestive tract. After mid-gestation, endothelial differences across organs became more pronounced, with increasing activation of organ-specific gene programs and signaling pathways that aligned with each organ’s physiological roles.
This atlas facilitated identification of unknown lung endothelium enriched gene such as Casz1.
Endothelial-specific deletion of Casz1 impaired lung vascular growth and prevented lung endothelial cells from acquiring organ-specific characteristics. Mechanistically, Casz1 binds promoter regions in lung endothelial chromatin and influences expression of essential genes for lung vascular development and maturation, including Vegfr2, Foxf1, and Car4.
Loss of Casz1 in endothelial cells also reduced NKX2.1+ and SFTPC+ alveolar epithelial cells, delayed their proliferation, and weakened endothelial–epithelial cell interactions. The authors find these effects were associated with reduced endothelial release of FGF1, suggesting that Casz1 supports alveolar development in part by controlling endothelial paracrine signaling.
Overall, this work provides an endothelial developmental atlas across 26 embryonic time points and identifies the timing and molecular features of organ-specific endothelial cells. Using the lung as a model, it also connects organ-specific endothelial maturation to local tissue development through endothelial–epithelial signaling. These findings provide a foundation for studying how organ-specific vascular features contribute to development and diseases.
Contacts:
CHEN Qi, Ph.D., Principal Investigator;
Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences, Guangzhou, China, 510530.
Email: chen_qi@gibh.ac.cn
Attachment Download:
-
ContactCHEN Qi, Ph.D.chen_qi@gibh.ac.cn
-
Reference
-
Related Article