Researchers Develop a Safer and More Efficient mTOR Inhibitor
Recently, a research team led by Prof. TANG Shibing from the Guangzhou Institutes of Biomedicine and Health (GIBH), Chinese Academy of Sciences, published a research paper titled "Discovery and Optimization of 4-Aminopteridin-7(8H)-one Derivatives as Potent and Selective mTOR Inhibitors with Favorable Pharmacodynamic and Safety Characteristics" online in the Journal of Medicinal Chemistry. The paper reports a novel class of mTOR inhibitors with excellent efficacy and safety profiles.
The mTOR (mammalian target of rapamycin) signaling pathway is a central hub regulating cell growth, proliferation, and survival. Its abnormal activation is closely associated with the development and progression of various cancers such as gastric cancer, lung cancer, and breast cancer, making it a crucial target for anti-cancer drug development. However, currently available mTOR inhibitors on the market remain very limited, with the vast majority of candidate drugs failing during clinical development due to insufficient potency, excessive toxicity, or severe side effects. Therefore, developing a new generation of mTOR inhibitors that are both potent and safe is an urgent challenge to overcome in this field.
Addressing this challenge, the GIBH research team successfully developed a class of 4-aminopteridin-7(8H)-one derivatives as ATP-competitive mTOR inhibitors through rational drug design. Among them, the lead compound T133 (compound 51) demonstrated particularly outstanding performance. Through structure-activity relationship studies and molecular simulations, the research revealed that the hydroxylated benzofuran and trans-cyclohexanecarboxylic acid fragments in the T133 molecule confer high affinity (IC₅₀ = 0.34 nM) and good kinase selectivity by forming specific hydrogen bonds and salt bridge interactions with mTOR.
Cellular experiments showed that T133 effectively inhibited cell proliferation and migration across various cancer cell lines, including gastric, lung, and breast cancer. Mechanistic studies further confirmed that T133 blocks the mTOR downstream signaling pathway, significantly reducing the phosphorylation levels of key signaling proteins such as AKT, S6K1, and 4EBP1, thereby inducing tumor cell apoptosis, cell cycle arrest, and autophagy.
In the HGC-27 gastric cancer cell xenograft model, oral administration of T133 exhibited dose-dependent tumor inhibition effects: tumor inhibition rates reached 83% and 92% at doses of 30 mg/kg and 60 mg/kg, respectively. Its efficacy was comparable to the clinically investigated inhibitor PF-04691502, but T133 showed significant advantages in safety. Unlike PF-04691502, which caused liver damage, lung injury, skin toxicity, and other adverse effects consistent with clinical reports, mice treated with T133 showed no significant pathological damage in vital organs, and their physiological and biochemical indicators remained stable. Furthermore, the broad-spectrum anti-cancer effect of this drug was also validated in the NCI-H1299 lung cancer model.
Regarding drug-like properties, T133 exhibits good oral bioavailability (31.2%), shows very weak inhibition of major drug-metabolizing enzymes (CYP450), has no potential cardiotoxicity (extremely low hERG inhibition), and is non-genotoxic (AMES test negative), indicating an overall low drug development risk.
In summary, based on unmet clinical needs and focusing on the toxicity bottlenecks of existing mTOR inhibitors, this study achieved the candidate molecule T133 through rational structural optimization. While maintaining high potency and selectivity, T133 demonstrated excellent safety characteristics in preclinical studies, bringing new hope for overcoming the development challenges of second-generation mTOR inhibitors.
This research was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences, the Guangdong Basic and Applied Basic Research Foundation, and the Basic Research Project of Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences.
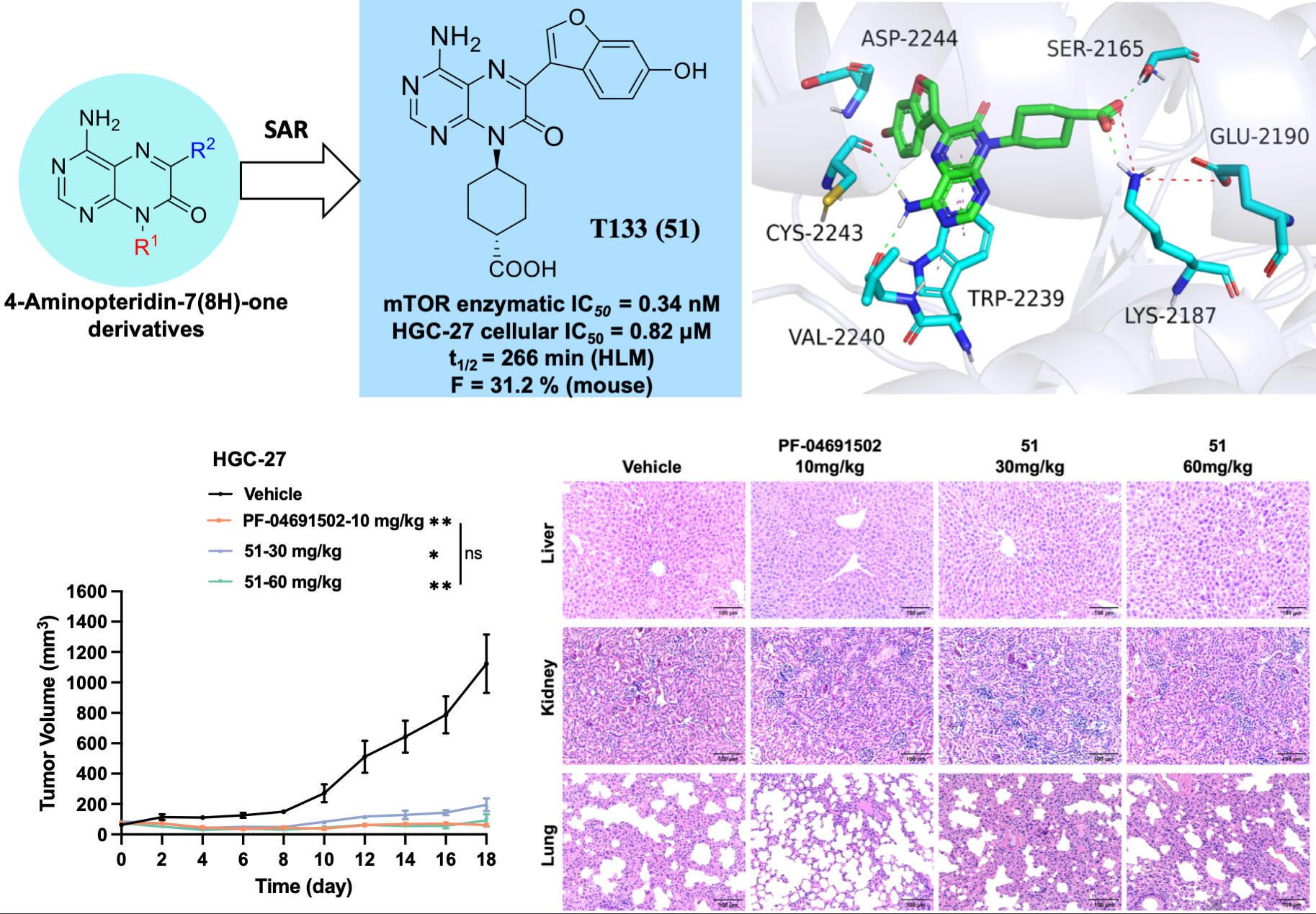
Figure 1. Rational drug design and optimization lead to the development of a safer and more efficient novel mTOR inhibitor.
Contacts:
TANG Shibing, Ph.D., Principal Investigator
Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences, Guangzhou, China, 510530
Email: tang_shibing@gibh.ac.cn
Attachment Download:
-
ContactTANG Shibing, Ph.D.tang_shibing@gibh.ac.cn
-
Reference
-
Related Article