Researchers Reveal "Domesticated" Transposon Protein as the Gatekeeper of Pluripotency
A recent study from LIU Xingguo’s group at the Guangzhou Institutes of Biomedicine and Health (GIBH), Chinese Academy of Sciences, was published in Cell Discovery, titled “Post-transcriptional control of endogenous retroviruses by L1td1 suppresses totipotency acquisition in pluripotent stem cells.” The work reports that L1td1, a protein evolutionarily co-opted from the LINE-1 retrotransposon, acts as a critical "gatekeeper" that restricts pluripotent stem cells (PSCs) from reverting to a totipotent state. The study demonstrates that loss of L1td1 leads to the reactivation of totipotency-associated genes and endogenous retroviruses (ERVs), causing cells to spontaneously regress to a totipotent-like state (or 2-cell-like state) resembling the earliest stages of embryogenesis. This study identifies L1td1 as a key post-transcriptional regulator that suppresses endogenous viral elements to maintain pluripotency.
The transition from totipotency to pluripotency is a fundamental event in early embryonic development, characterized by the silencing of totipotent genes and specific retrotransposons activated during zygotic genome activation (ZGA). While transcriptional and epigenetic mechanisms governing this developmental restriction have been extensively studied, the role of post-transcriptional regulation, specifically RNA decay, has remained elusive. L1td1 is the only mammalian protein-coding gene domesticated from the "jumping" LINE-1 retrotransposon and is highly expressed in PSCs, yet its precise function in controlling cell fate transitions was previously unclear.
Using mouse and human PSC models, the authors demonstrate that L1td1 plays a decisive role in preventing the acquisition of totipotency. Mechanistically, L1td1 recruits the CCR4-NOT deadenylase complex to specifically target and degrade RNA transcripts essential for totipotency, including the Zscan4 gene family and ERVs such as MERVL-int and MT2_Mm. Notably, the study found that L1td1 also targets the RNA of its own evolutionary progenitor, LINE-1, for degradation. By clearing these transcripts, L1td1 effectively blocks the reversion to a 2-cell-like (2C-like) state, thereby securing the pluripotent identity of the cell.
Together, this work reveals a previously unrecognized mechanism wherein a co-opted transposon protein utilizes the CCR4-NOT machinery to bridge transposon silencing with cell fate determination. It highlights the importance of RNA stability control in early embryonic development and genome defense. These findings provide new theoretical insights into the evolutionary interplay between viral elements and host developmental regulation, offering potential strategies for inducing totipotency in regenerative medicine and xenogeneic organ regeneration.
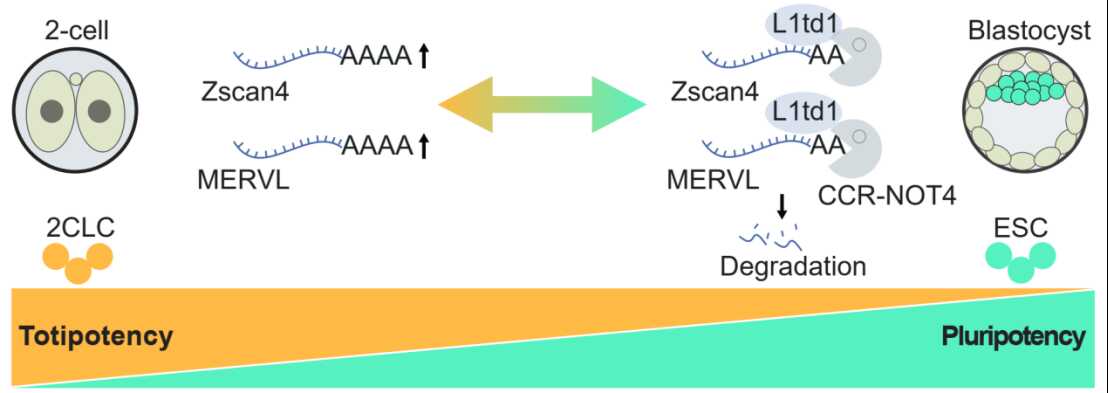
Image by Prof. LIU's team.
This study was supported by the National Key R&D Program of China, the Chinese Academy of Sciences, the National Natural Science Foundation of China, and funding from Guangdong Province and Guangzhou City. The work was led by Prof. LIU Xingguo and Dr. WU Yi (co-corresponding authors), with WU Yi, LIU Yang, and HUANG Yile serving as co-first authors.
Contacts:
LIU Xingguo, Ph.D., Principal Investigator;
Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences, Guangzhou, China, 510530.
Email: liu_xingguo@gibh.ac.cn
Attachment Download:
-
ContactLIU Xingguo, Ph.Dliu_xingguo@gibh.ac.cn
-
Reference
-
Related Article