International Collaboration Uncovered Potential Therapeutic Strategies for Solid Tumors by Targeting UHRF1 for DNA Demethylation
A recent study, led by Dr. KONG Xiangqian, Dr. LIU Jinsong, and Dr. WU Linping's labs at the Guangzhou Institutes of Biomedicine and Health (GIBH), Chinese Academy of Sciences (CAS), in collaboration with Dr. Stephen Baylin's group at The Johns Hopkins University School of Medicine (JHMI), was published in Nature Communications.The paper is titled "Defining ortholog-specific UHRF1 inhibition by STELLA for cancer therapy."
Building upon earlier findings by Dr. KONG and Dr. Baylin (Cancer Cell, 2019), this study explores the species-specific inhibitory effects of the pluripotency regulator STELLA on UHRF1, an oncogenic driver essential for maintaining abnormal DNA hypermethylation in human cancers. These findings prompted the development of an mRNA-based UHRF1 inhibitor that effectively reverses cancer-specific DNA methylation and impairs colorectal cancer tumorigenicity.
The study found that human STELLA (hSTELLA) is significantly less effective than mouse STELLA (mSTELLA) in disrupting UHRF1’s chromatin association and nuclear localization. Structural studies revealed a region of low sequence homology between these STELLA orthologs, allowing mSTELLA but not hSTELLA to bind tightly and cooperatively to the essential histone-binding TTD-PHD domain of UHRF1, thus mediating ortholog-specific UHRF1 inhibition. For translating these findings into cancer therapy, the researchers developed a lipid nanoparticle (LNP)-mediated mRNA delivery strategy. They demonstrated that delivering mRNA encoding an “L-shaped” binding conformation of mSTELLA significantly reduces DNA hypermethylation and leads to durable tumor growth inhibition through an epigenetic memory effect. In contrast, delivering mRNA encoding the “long α-helix” conformation of hSTELLA failed to exhibit such effects.
Dysregulated UHRF1 functions are associated with poor patient prognosis, making it a promising target for cancer treatment. Despite growing interests in developing UHRF1 inhibitors, no small molecule or peptide inhibitors have yet been reported to target UHRF1 for reversing genome-wide DNA methylation abnormalities in cancer cells. This study not only lays the foundation for developing mRNA-based DNA demethylation agents but also suggests the potential for rationally developing peptidomimetic or small molecule mimics of mSTELLA.
Dr. BAI Wenjing, Dr. XU Jinxin, Dr. GU Wenbin, and Dr.WANG Danyang from GIBH, along with Dr. CUI Ying from JHMI, are the co-first authors of this paper. The project was jointly supervised by Dr. KONG, Dr. LIU, and Dr. Baylin, with significant contributions from Dr. WU of GIBH, CAS and Dr. LUO Cheng of the Shanghai Institute of Materia Medica, CAS. The authors gratefully acknowledge the financial support from the National Natural Science Foundation of China, the Basic Research Project of Guangzhou Institutes of Biomedicine and Health, Guangdong Basic and Applied Basic Research Foundation, National Key R&D Program of China, and others.
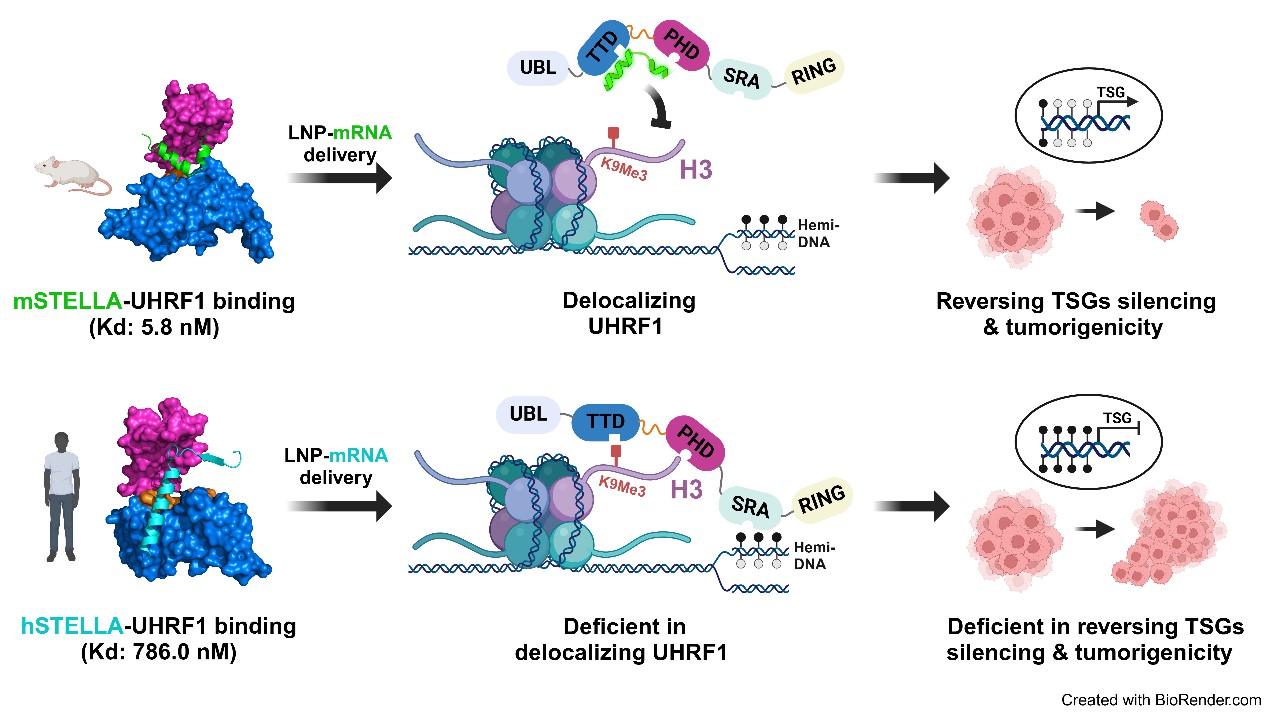
Fig. Ortholog-specific effects of mSTELLA and hSTELLA in inhibiting the abnormal DNA methylation and oncogenic functions of UHRF1 in human cancer cells (Image by Prof. KONG's team)
Contacts:
KONG Xiangqian, Ph.D., Principal Investigator;
Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences, Guangzhou, China, 510530.
Email: kong_xiangqian@gibh.ac.cn
Attachment Download:
-
ContactKONG Xiangqian, Ph.D.kong_xiangqian@gibh.ac.cn
-
Reference
-
Related Article